AI-powered visual inspection for pharmaceutical manufacturing
DeepInspect®- Pharmaceutical quality control automation using vision AI
DeepInspect® enables AI-powered visual quality inspection for pharmaceutical manufacturers with high-accuracy automated defect detection at production speed. From vial visual inspection and ampoule inspection to blister pack inspection and pharma bottle inspection, advanced deep learning algorithms identify surface defects, foreign particles, sealing integrity failures, OCV/OCR errors, and label mismatches in real time.
Built as an FDA 21 CFR Part 11-compliant software, DeepInspect® ensures secure audit trails, electronic record integrity, role-based access control, and complete traceability for regulated pharmaceutical environments. This simplifies validation, documentation, and regulatory audits.
By implementing AI-based pharmaceutical inspection systems, manufacturers achieve 100% inline inspection, reduced false rejection rates, improved packaging quality control, and enhanced compliance—safeguarding product quality and patient safety across pharma production lines.
Contact Form

Contact Form
USECASE
Detect surface defects in pharmaceutical products to ensure regulatory compliance
Maintain packaging integrity by identifying contamination and cracks in pharma products
Accurately detect particles, scratches, and micro-cracks during vial visual inspection
Prevent hairline fractures and air bubble defects in ampoules before dispatch
Eliminate leakage, misprints, and cavity defects in blister packaging lines
Verify cap fitment and seal integrity to avoid leakage and tampering risks
Ensure sealing integrity to prevent contamination, leakage, and sterility failures
Validate OCV/OCR accuracy to eliminate print errors and serialization mismatches
Inspect label & artwork quality to avoid regulatory non-compliance
SwitchOn in the News

AI-powered quality inspection for precision manufacturing
AI-powered computer vision based Quality inspection platform

AI-powered computer vision based Quality inspection platform

SwitchOn enables automotive and CPG discrete manufacturing industries to ensure product quality and process continuity.

Community Voices: How SwitchOn is enabling manufacturing companies reach zero-defects

AI-based vision inspection startup SwitchOn raises $4.2 million
SwitchOn's founders on how they're helping ITC, Unilever and SKF automate quality checks on the go
DeepInspect® – The best vision AI software for automated defect detection in pharma production lines
Deep learning visual inspection for pharma industry with 99.5% accuracy
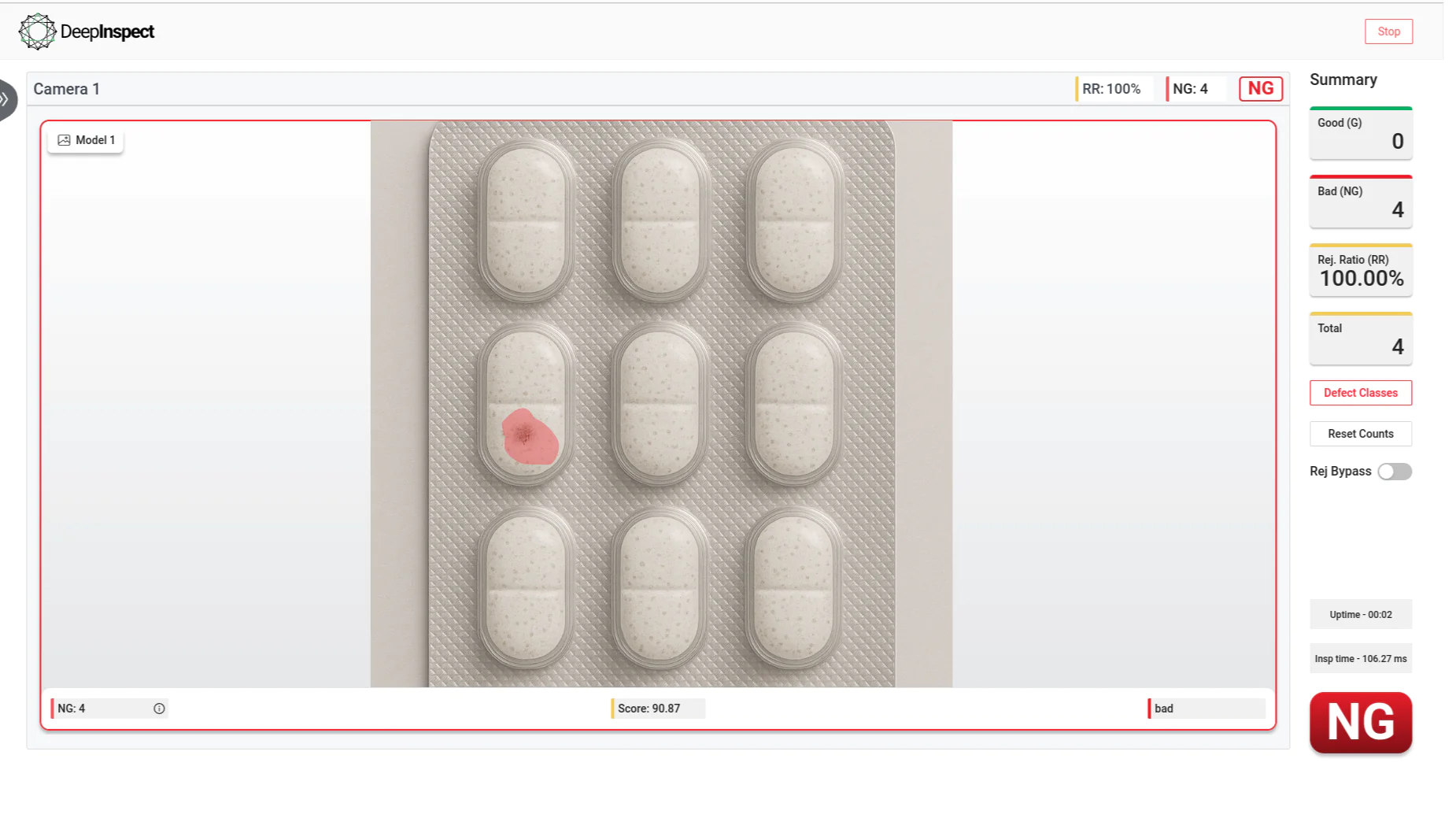
FDA 21 CFR Part 11-compliant visual inspection system for pharma manufacturers
Contact Form
False rejection reduction in pharma with print and label inspection

Why DeepInspect®
99.5% Inspection Accuracy: DeepInspect® delivers consistent, high-speed inspection accuracy of 99.5%, setting new benchmarks for reliability in automated quality control
Less Than 200 Good Images for Model Training: DeepInspect® requires fewer than 200 good images to learn, adapt, and begin accurate inspection with minimal setup effort
Model Training in Under 45 Minutes: DeepInspect® enables model creation and fine-tuning in under 45 minutes, reducing downtime and maximizing productivity
Line Trial in One Day: A complete line trial can be conducted at your plant within a single day
1000+PPM: High-speed inspection, detecting 1000+ parts per minute
Top vision inspection system for pharmaceutical industry

Our Customers
Trusted by the world’s leading manufacturers to power zero-defect production
Regulatory-compliant quality system for pharma companies
Interested in AI-powered pharma inspection?
Book a free demo!
Inquiry Form
Visual quality inspection in pharmaceutical manufacturing involves automated defect detection of surface defects, foreign particles, sealing failures, and label errors to ensure product safety and regulatory compliance.
Automated visual inspection reduces human error, ensures 100% inline inspection, improves defect detection accuracy, and minimizes product recalls in pharmaceutical production lines.
AI-based pharmaceutical inspection systems detect surface cracks, cosmetic defects, contamination, sealing integrity issues, OCV/OCR errors, label mismatches, and packaging defects.
AI-powered visual inspection enables real-time defect detection, reduces false rejections, enhances traceability, and ensures consistent pharmaceutical packaging quality.
DeepInspect® is one of the leading AI-powered visual inspection software solutions for pharmaceutical manufacturers, offering high-accuracy defect detection and regulatory-ready compliance features.
Yes, DeepInspect® is FDA 21 CFR Part 11-compliant software with secure audit trails, electronic record integrity, and role-based access control for regulated pharma environments.
DeepInspect® supports vial visual inspection, ampoule inspection, blister pack inspection, pharma bottle inspection, and cap and seal inspection across pharmaceutical production lines.
DeepInspect® delivers high defect detection accuracy with reduced false rejection rates, enabling reliable automated optical inspection for pharma manufacturers.
Yes, DeepInspect® validates OCV/OCR accuracy, detects serialization mismatches, and inspects label and artwork defects in pharmaceutical packaging.
DeepInspect® ensures complete inspection traceability, secure electronic records, and streamlined validation processes—strengthening regulatory compliance and product quality assurance.